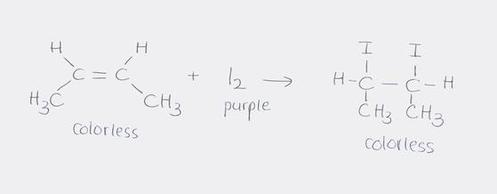B.4.1 Compare the composition of of the three types of lipids found in the human body
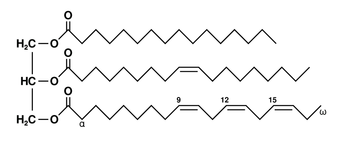
Triglyceride (fats and oils):
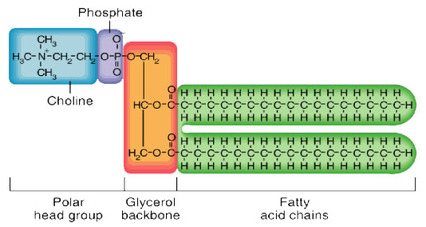
Phospholipid:
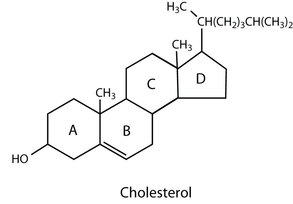
Steroid:
Triglyceride (fats and oils):
- triglycerides are formed through an ester linkage between a glycerol molecule (propan-1,2,3-triol) and 3 fatty acid molecules
- fats are solid at room temperature and oils are liquid
- low solubility in water, soluble in non-polar solvents
- 1) simple triglyceride: all 3 fatty acids are the same
- 2) mixed triglyceride: different fatty acids attached to the same glycerol
Phospholipid:
- composed of 2 fatty acids attached to a glycerol, which is attached to a phosphate bonded to a polar alcohol
- general structure is a polar/charged head and non-polar tails
- cell membranes are phospholipid bilayers
- proteins are associated with or embedded in the bilayer
Steroid:
- steroids contain a tetracyclic fused carbon ring
- composed of 3 cyclohexane derivatives fused to 1 cyclopentane derivative
- different steroids have different functional groups
- cholesterol is the most abundant and most important steroid because they're the basis for synthesis of many hormones and vitamins, and it helps maintain cell membrane fluidity
B.4.2 Outline the difference between HDL and LDL cholesterol and outline its importance
- HDL - high density lipoprotein
- LDL - low density lipoprotein
- both transport lipids (which are not water soluble) through the blood
- HDL can absorb more cholesterol than LDL and can remove it from problem sites
- LDL carries cholesterol to sites to build and repair within the body, but a high LDL level tends to result in cholesterol accumulating at problem sites in the body (e.g. atherosclerotic plaques)
B.4.3 Describe the difference in structure between saturated and unsaturated fatty acids
Saturated Fats:
Unsaturated Fats:
Note: Most naturally occurring fats contain a mixture of saturated, mono-unsaturated, and poly-unsaturated fatty acids and are classified according to the predominant type of unsaturation present
Saturated Fats:
- all carbon-carbon bonds are single
- common in most animal fats
- solid at room temperature
- they pack closely, so although London forces are weak, the R groups interact enough to form solids
Unsaturated Fats:
- at least one double C=C bond in at least one chain
- monounsaturated - one C=C double bond per chain
- polyunsaturated - more than one C=C double bond per chain
- liquids at room temperature
- double bond(s) prevent molecules from packing closely enough to have strong enough London forces to be a room temperature solid
- naturally occurring C=C bonds in fats are cis isomers
Note: Most naturally occurring fats contain a mixture of saturated, mono-unsaturated, and poly-unsaturated fatty acids and are classified according to the predominant type of unsaturation present
B.4.4 Compare the structures of the two essential fatty acids, linoleic (omega-6 fatty acid) and linolenic (omega-3 fatty acid) and state their importance
- these are called essential because the body can't synthesize them and they have to be obtained from your diet
- a lack of either can cause ill health and deficiency symptoms
- linoleic acid (C18H32O2) has 2 C=C double bonds, while linolenic acid (C18H30O2) has 3 C=C double bonds
B.4.5 Define the term iodine number and calculate the number of C=C double bonds in an unsaturated fat/oil using addition reactions
E.g.) 0.010 mol linoleic acid reacts with 5.1 g of iodine (I2). Determine the number of double bonds present in linoleic acid.
5.1 g I2 x (1 mol I2 / 253.8 g I2) x (1 mol C=C / 1 mol I2) = 0.020 mol C=C
(0.020 mol C=C)/(0.010 mol linoleic acid) = 2 C=C per linoleic acid
- iodine number is the measure of the degree of unsaturation of an oil, fat, or wax - the amount of iodine in grams that is taken up by 100 grams of the oil, fat, or wax
E.g.) 0.010 mol linoleic acid reacts with 5.1 g of iodine (I2). Determine the number of double bonds present in linoleic acid.
5.1 g I2 x (1 mol I2 / 253.8 g I2) x (1 mol C=C / 1 mol I2) = 0.020 mol C=C
(0.020 mol C=C)/(0.010 mol linoleic acid) = 2 C=C per linoleic acid
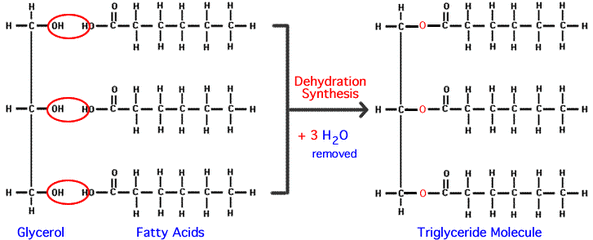
B.4.6 Describe the condensation of glycerol and three fatty acid molecules to make a triglyceride
- 3 hydrogens from a glycerol molecule and hydroxyl groups from 3 fatty acids detach to form 3 molecules of water as a byproduct
- ester linkages are formed between the oxygens on the glycerol molecule and the carbonyl groups on the fatty acids
B.4.7 Describe the enzyme-catalyzed hydrolysis of triglycerides during digestion
- reverse of esterification
- catalyzed by enzyme lipase
- reaction is stepwise (each R group removed sequentially)
- triglyceride + 3H2O --> glycerol + 3 fatty acids
- triglyceride --> diglyceride --> monoglyceride --> glycerol
B.4.8 Explain the higher energy value of fats as compared to carbohydrates
- fats have fewer oxygen than carbohydrate molecules of corresponding molar mass, thus more oxidation can take place
- therefore, more energy is released from the oxidation of fats as opposed to carbohydrates
- fats provide nearly twice as much energy (kJ/g) as carbohydrates
B.4.9 Describe the important roles of lipids in the body and the negative effects that they can have on health
Important Roles Include:
Negative Effects Include:
Important Roles Include:
- energy storage
- insulation and protection of organs
- steroid hormones
- structural component of cell membrane
- omega-3 poly-unsaturated fatty acids reduce the risk of heart disease
- poly-unsaturated fats may lower levels of LDL cholesterol
Negative Effects Include:
- increased risk of heart disease from elevated levels of LDL cholesterol and trans fatty acids; the major source of LDL cholesterol is saturated fats, in particular lauric (C12), myristic (C14), and palmitic (C16) acids
- potential obesity