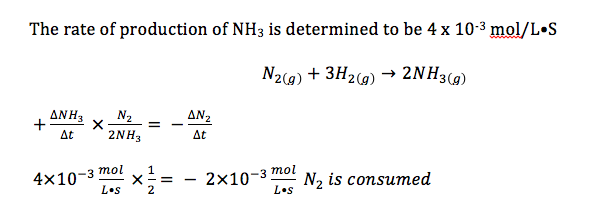6.1.1 Define the term rate of reaction
- rate of reaction: the amount of reactant that is converted to product (or product produced from reactant) per unit of time
6.1.2 Describe suitable experimental procedures for measuring rates of reaction
- e.g.) CaCO3 + 2HCl --> CaCl2 + CO2 + H2O
- could measure gas produced (CO2), pH (acidic to nonacidic), mass change (if environment is open and CO2 leaves the system), or mass change of CaCO3
6.1.3 Analyze data from rate experiments
E.g.) Determine the rates of reaction for each of the other reactants in the following reaction:
- rate is always expressed as a value per unit of time
- the initial rate is usually considered to be the most accurate
- the numerical value of the reaction rate is always positive
- a + or - is merely used to show whether one is measuring the production or consumption of a product or reactant
- rates of formation of products and consumption of reactants are related by stoichiometry
E.g.) Determine the rates of reaction for each of the other reactants in the following reaction: