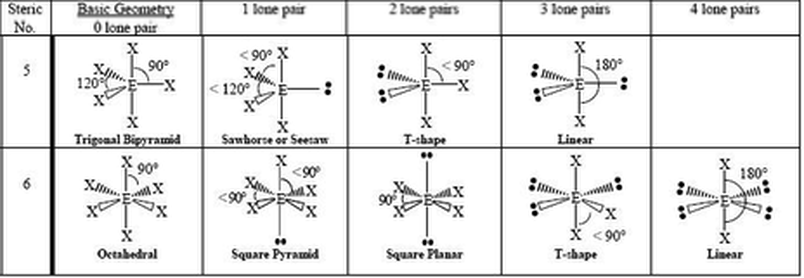
14.1.1 Predict the shape and bond angles for species with 5 and 6 negative charge centers using the VSEPR theory
Principles of VSEPR:
5 and 6 Charge Centers:
Principles of VSEPR:
- find the # of charge centers in the valence shell of the central atom (do not neglect Lewis dot diagram)
- electron pairs/groups around the central atom repel each other (not atoms or bonds)
- these groups are called charge centers
- electron pairs are found in positions of minimum energy/maximum stability, so as a whole, charge centers will be furthest apart
- double or triple bonds act like single bonds/are a single charge center
- the magnitude of repulsion is greatest between a lone pair and lone pair, and smallest between bonding pair and bonding pair
5 and 6 Charge Centers:
- octet rule is ignored
- atoms that can possibly have 4+ charge centers: P, S, Cl, Br, I, Xe
- all central atoms involved must have electrons in d orbitals (they're involved in bonding)