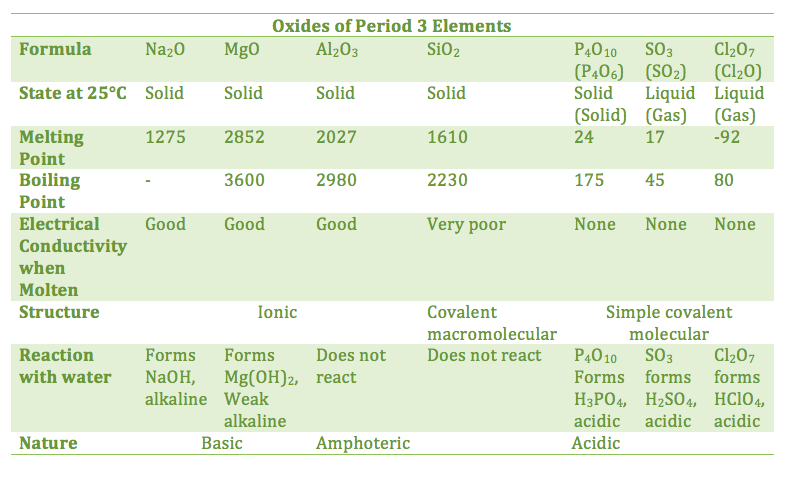
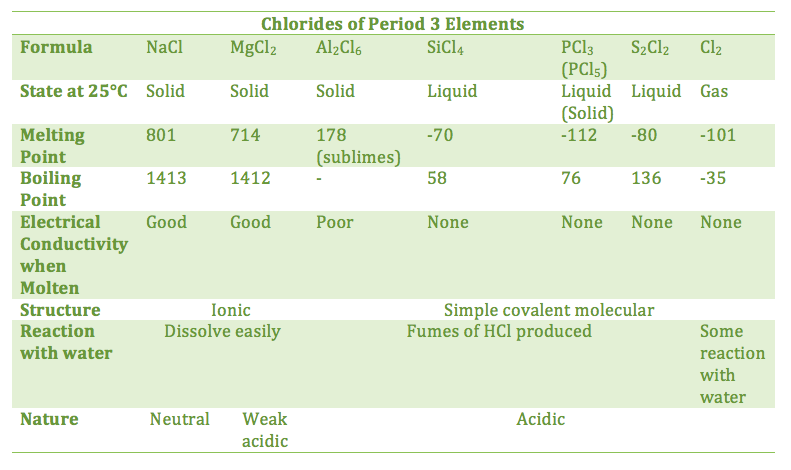
13.1.1 Explain the physical states (under standard conditions) and electrical conductivity (in the molten state) of the chlorides and oxides of the elements in period 3 in terms of their bonding and structure
- sodium, magnesium, and aluminum oxides are ionic
- thus, they have high melting points and electrical conductivity when molten
- silicon dioxide is a macromolecular covalent structure (diamond-like) with a high boiling point
- non-metallic oxides have a lower difference in electronegativity with oxygen, and form simple molecular covalent structures, resulting in low melting and boiling points
13.1.2 Describe the reactions of chlorine and the chlorides referred to in 13.1.1 with water
- physical properties of chlorides similar to oxides
- sodium and magnesium chlorides are ionic, conduct electricity when molten, and have high melting points
- aluminum chloride is covalent and a poor conductor
- silicon tetrachloride has a simple molecular structure like the remaining chlorides, which are held by weak Van de Waal's forces and thus result in low melting and boiling points
- sodium chloride dissolves in water to give a neutral solution, while magnesium chloride gives a slightly acidic solution
- all other chlorides (including aluminum) react with water to produce acidic solutions of HCl with fumes of hydrogen chloride
- chlorine itself reacts with water to some extent to form an acidic solution