2.2.1 Describe and explain the operation of a mass spectrometer
5 Stages
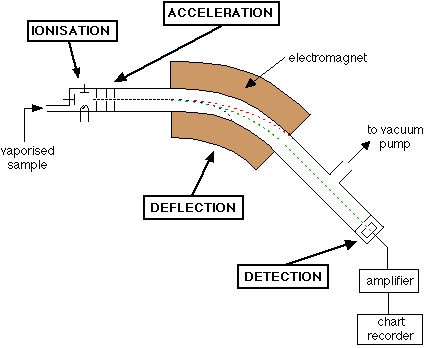
1) Vapourization: Particles are vapourized and kept under very low pressure. Heat/electricity is used because individual particles must strike detector so that ions (particles) do not interact with each other. Generally solids or liquids are heated.
Capillary Tube: Gases pass through this slowly (small diameter). This combined with the low pressure of the vacuum will give individual particles.
2) Ionisation: Particles are converted from neutral into positive ions, usually by fast moving stream of electrons. The electron stream knocks electrons off of particles entering the device. Since the mass spectrometer is usually calibrated for a single charge, it's usually 1+ (1 electron knocked off).
3) Acceleration: Ions are accelerated by passing through a pair of high voltage (potential difference) electrodes/plates with holes in the center. All particles are accelerated to the same velocity.
4) Deflection: Fast moving ions enter magnetic field established perpendicularly to the direction of motion of charged particles. Ions are deflected in an arc. The radius of deflection is proportional to the charge/mass ratio of ions. Only particles of one particular mass will continue around the curved tube and hit the detector plate at any given setting. By varying the strength of the magnetic field, ions of different mass can be focused progressively on the detector at the end of the tube.
5) Detection: Ions hit the metal plate which gives up electrons, creating an electric current. The current is amplified and sent to the recorder (computer). Ions that don't hit the plate are harmless and neutralized by the glass tube and sucked out by the vacuum.
5 Stages
1) Vapourization: Particles are vapourized and kept under very low pressure. Heat/electricity is used because individual particles must strike detector so that ions (particles) do not interact with each other. Generally solids or liquids are heated.
Capillary Tube: Gases pass through this slowly (small diameter). This combined with the low pressure of the vacuum will give individual particles.
2) Ionisation: Particles are converted from neutral into positive ions, usually by fast moving stream of electrons. The electron stream knocks electrons off of particles entering the device. Since the mass spectrometer is usually calibrated for a single charge, it's usually 1+ (1 electron knocked off).
3) Acceleration: Ions are accelerated by passing through a pair of high voltage (potential difference) electrodes/plates with holes in the center. All particles are accelerated to the same velocity.
4) Deflection: Fast moving ions enter magnetic field established perpendicularly to the direction of motion of charged particles. Ions are deflected in an arc. The radius of deflection is proportional to the charge/mass ratio of ions. Only particles of one particular mass will continue around the curved tube and hit the detector plate at any given setting. By varying the strength of the magnetic field, ions of different mass can be focused progressively on the detector at the end of the tube.
5) Detection: Ions hit the metal plate which gives up electrons, creating an electric current. The current is amplified and sent to the recorder (computer). Ions that don't hit the plate are harmless and neutralized by the glass tube and sucked out by the vacuum.
2.2.2 Describe how the mass spectrometer may be used to determine relative atomic mass using the carbon-12 scale
Mass Spectrum:
- mass spectrometer is an instrument that separates and detects charged particles
- particles with the same charge to mass ratio follows the same path through the instrument
- spectrometer can determine the abundance of each isotope and relative atomic mass of an element
Mass Spectrum:
- relative abundances of ions from the sample can be determined, often displayed graphically
- interpret by comparing to value of field required to deflect carbon-12
2.2.3 Calculate non-integer relative atomic masses and abundance of isotopes from given data
Relative Atomic Mass (Ar):
Relative Atomic Mass (Ar):
- the mass of the average atom of an element, relative to an atom of carbon-12
- calculation of Ar takes into account different isotopes and natural isotopic abundances (proportions of different isotopes that occur in nature, usually expressed as %)