17.1.1 Describe the equilibrium established between a liquid and its own vapour and how it is affected by temperature changes
Vapour Pressure:
- liquid-vapour equilibrium is a dynamic equilibrium established when the rate of condensation equals the rate of evaporization
- fast-moving particles in the liquid will escape from the surface and become part of the vapour, but slow-moving particles in the vapour will be "captured" by the liquid and become part of it
Vapour Pressure:
- the pressure exerted by a vapour on its liquid
- vapour in the container collides with the liquid surface and exerts a pressure on it
- at equilibrium (or saturation), this measured pressure is the vapour pressure
- the magnitude of vapour pressure depends on equilibrium position
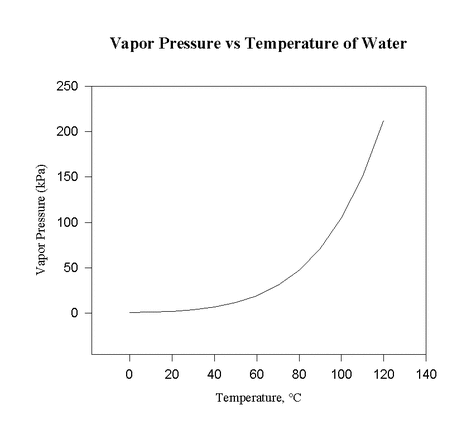
17.1.2 Sketch graphs showing the relationship between vapour pressure and temperature and explain them in terms of the kinetic theory
- as the temperature increases, the average speed of particles is higher
- as a result, more particles will have sufficient speed to escape the liquid, and fewer will be slow enough to fall back down to liquid
- thus, as temperature increases, equilibrium vapour pressure will also increase
17.1.3 State and explain the relationship between enthalpy of vapourization, boiling point, and intermolecular forces
- at the point where vapour pressure = the external pressure, the boiling point is reached and vapourization becomes a "volume phenomenon" (phase change is occurring throughout the liquid instead of just at the surface)
- liquids with high boiling points have strong intermolecular forces
- enthalpy of vapourization is a measure of the energy change when 1 mol of liquid is converted to gas at standard pressure
- thus, a lower enthalpy of vapourization means that less energy is required to break the intermolecular forces and thus result in a higher vapour pressure