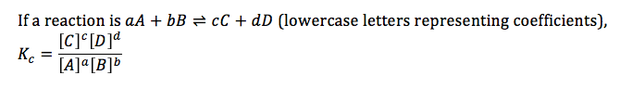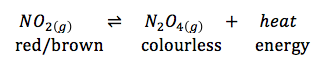7.2.1 Deduce the equilibrium constant expression (Kc) from the equation for a homogeneous reaction
- the equilibrium constant (Kc) expresses the relationship between products and reactants of a reaction at equilibrium with respect to a specific unit
- Kc values can be determined for both homogeneous and heterogeneous mixtures
- homogeneous mixtures are when all the reactants and products are in the same state
- heterogeneous mixtures are when the reactants and products are of different states
- the importance to remembering this is because any reactants and products that are solid (s) or pure liquid (l) must be excluded from the Kc formula (only concentrations of gaseous and aqueous reactants/product are used when calculating Kc)
- to find Kc for a homogeneous, find the concentrations of the reactants and products, substitute the corresponding values in along with the balanced equation's coefficients as the exponents and then multiply/divide
7.2.2 Deduce the extent of a reaction from the magnitude of the equilibrium constant
- the extent of completion of a reaction (eg. R <--> P) can be determined from the equilibrium constant
- Kc very large (105), reaction effectively goes to completion
- Kc > 1 (10-1000), reaction is product favoured although both forward and reverse reactions are measurable
- Kc ~ 1 (1-10), forward and reverse reactions are same magnitude
- Kc < 1 (1/10 - 1/1000), reactant-favoured although both forward and reverse reactions are measurable
- Kc very small, reaction effectively does not happen
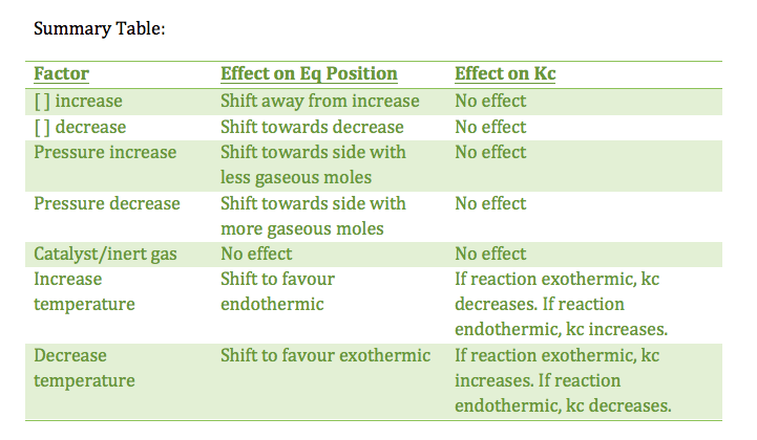
7.2.3 Apply Le Chatelier's principle to predict the qualitative effects of changes of temperature, pressure, and concentration on the position of equilibrium and on the value of the equilibrium constant
Le Chatelier's Principle (not required to define for exam): If a change is made to the conditions of a chemical equilibrium then the position of equilibrium will readjust so as to minimize the change made.
Temperature as a Stress:
Le Chatelier's Principle (not required to define for exam): If a change is made to the conditions of a chemical equilibrium then the position of equilibrium will readjust so as to minimize the change made.
Temperature as a Stress:
- adding energy/heat will cause the equilibrium position to shift in favour of the endothermic reaction
- need to "use up" the extra input of energy by converting it to potential energy
- conversely, removing energy/heat causes the equilibrium position to shift in favour of the exothermic reaction
- for the reaction above, adding ice makes the colour lighter because a decrease in heat favours the exothermic reaction (the forward reaction in this case), which will cause more colourless N2O4 to be produced
- adding heat makes the colour darker because the endothermic reaction is favoured (the reverse reaction in this case) and so not as much red/brown NO2 will be converted to product
Concentration Changes:
- with concentration changes, the equilibrium will shift to minimize the change in [ ]
- for a concentration increase, the system will shift away from the side where the increase occurred
- for a concentration, decrease, the system will shift towards the side where the decrease occurred
Gas Volume/Pressure Changes
- changes only apply if gases are present in the reaction
- a system will react to try to regain the original overall pressure
- increasing pressure of the system (decreasing volume) will cause the system to shift to the side with less gaseous moles
- decreasing pressure (increasing volume) will cause the system to shift to the side with more gaseous moles
7.2.4 State and explain the effect of a catalyst on an equilibrium reaction
- a catalyst decreases the time required to reach equilibrium (due to a decrease in activation energy)
- however, both the forward and reverse reactions are sped up, thus equilibrium position does not change
7.2.5 Apply the concepts of kinetics equilibrium to industrial processes
Contact Process: http://www.chemguide.co.uk/physical/equilibria/contact.html
Haber Process: http://www.chemguide.co.uk/physical/equilibria/haber.html
Contact Process: http://www.chemguide.co.uk/physical/equilibria/contact.html
Haber Process: http://www.chemguide.co.uk/physical/equilibria/haber.html