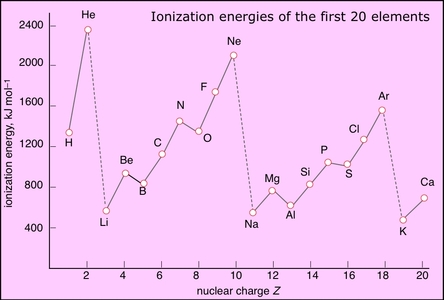
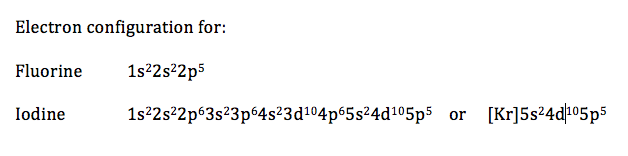12.1.1 Explain how evidence from first ionization energies across periods accounts for the existence of main energy levels and sub-levels in atoms
- first ionization energy of an element is the energy required to remove one electron from an atom in its gaseous state (measured in kJ/mol)
|
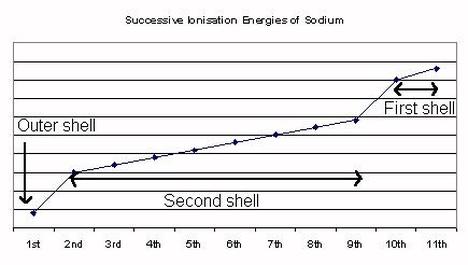
12.1.2 Explain how successive ionization energy data is related to the electron configuration of an atom
|
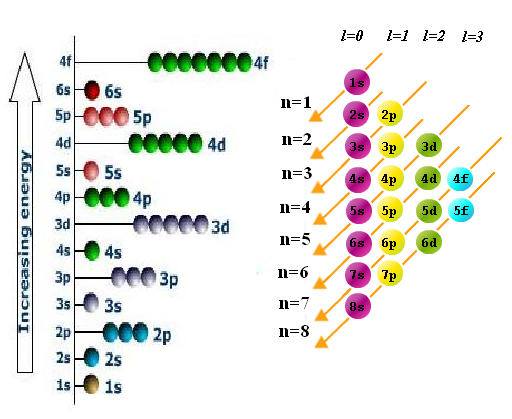
12.1.3 State the relative energies of s, p, d, f orbitals in a single energy level
- instead of electrons being found in strict shells around the nucleus like taught with the Bohr-Rutherford diagram, electrons are actually found in orbitals
- each orbital can contain a maximum of 2 electrons with opposite spin directions
- the first energy level contains 1 orbital, an s orbital (called 1s because it's in the 1st main level)
- the second energy level contains one s orbital (2s) and three p orbitals (2p)
- the three 2p orbitals have each the same amount of amount, but all have slightly higher energy than the 2s orbital
12.1.4 State the maximum number of orbitals in a given energy level
Main Energy Level/Shell # of each type of orbital Max. # electrons in main level
s p d f
1 1 - - - 2
2 1 3 - - 8
3 1 3 5 - 18
4 1 3 5 7 32
Main Energy Level/Shell # of each type of orbital Max. # electrons in main level
s p d f
1 1 - - - 2
2 1 3 - - 8
3 1 3 5 - 18
4 1 3 5 7 32
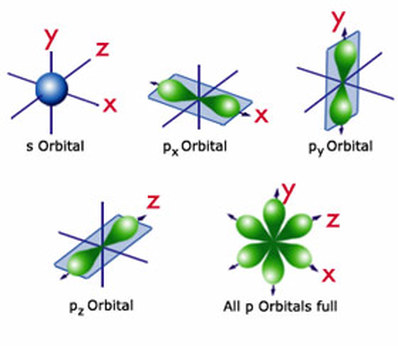
12.1.5 Draw the shape of an s orbital and the shapes of the px, py, and pz orbitals
- because an electron has the properties of both a particle and a wave, Heisenberg's uncertainty principle states that it's impossible to know the exact position of an electron at a precise moment in time
- the shape of an orbital shows the 3-D space where there is a high probability that the electron will be located
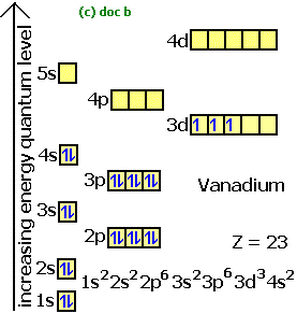
12.1.6 Apply the Aufbau principle, Hund's rule, and Pauli exclusion principle to write electron configurations for atoms and ions up to Z=54
- Aufbau principle: Orbitals with the lowest energy are filled first
- Hund's rule: Orbitals within the same sub-shell are filled singly first (e.g. one electron is put into each separate 2p orbital first so that they are each half-full instead of filling one of the 2p orbitals with 2 electrons and then moving onto the next 2p orbital)
- Pauli exclusion principle: No 2 electrons can have the same 4 quantum numbers (the 2 electrons in each full orbital would have to have opposite directions of spin, as spin is one of the quantum numbers)
- the superscript numbers represent the number of electrons in the s, p, or d sub-level
- configurations can be written out in full, or, to shorten it for elements with large atomic numbers, the name of the noble gas that preceded the element can be written (in square brackets) followed by electrons for only the orbitals after that noble gas
- in a neutral atom, the sum of the superscripts should be equal to that element's atomic number
- there are exceptions to this rule with the configurations of Cu and Cr
- instead of Cu and Cr having a full 4s orbital with 2 electrons, the 3d orbitals are so unstable because they're almost half-full (5 electrons) or full (10 electrons), that one of the 4s electrons is excited to join the 3d orbitals to make them preferentially half-full or full
- electron configurations are also often drawn in the form of a diagram by placing arrows in boxes, with the opposite arrow directions in each box showing how the 2 electrons within an orbital have opposite spins