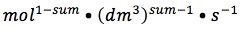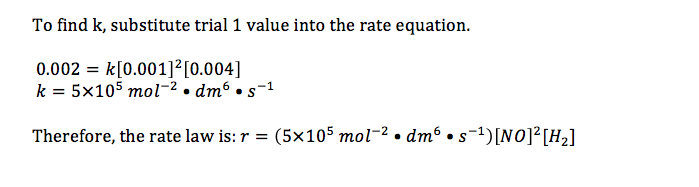16.1.1 Distinguish between the terms rate constant, overall order of reaction, and order of reaction with respect to a particular reactant
- the law of mass action: the rate of a chemical reaction (r) is proportional to the product of the concentrations of the reactants
- exponents in the rate law can only be determined empirically
- they can have any real number value (including integers, fractions, or zero)
- they do NOT have to equal the coefficients of the reactants in the balanced equation
Rate Constant (k):
- it's empirically determined
- this particular value is valid only for one specific chemical reaction
- k for a specific reaction is affected by temperature, but NOT affected by concentration changes
- units for k depend on the rate law
- general formula for determining units of k:
Individual Order of Reaction: The value of the exponent in the rate law associated with the concentration of a specific substance.
Overall Order of Reaction: The sum of all of the exponents in the rate law associated with concentrations of substances.
Overall Order of Reaction: The sum of all of the exponents in the rate law associated with concentrations of substances.
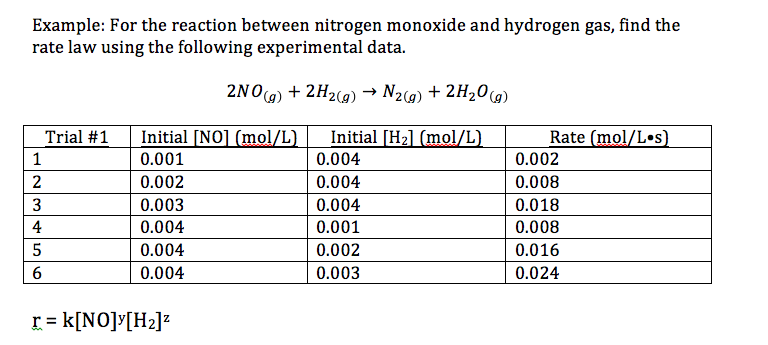
16.1.2 Deduce the rate expression for a reaction from experimental data
16.1.3 Solve problems involving the rate expression
16.1.4 Sketch, identify, and analyze graphical representations for zero, first, and second order reactions
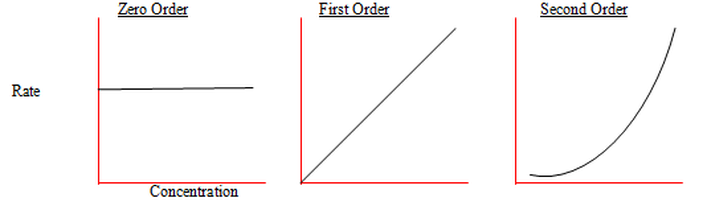
Concentration vs. Rate Graphs
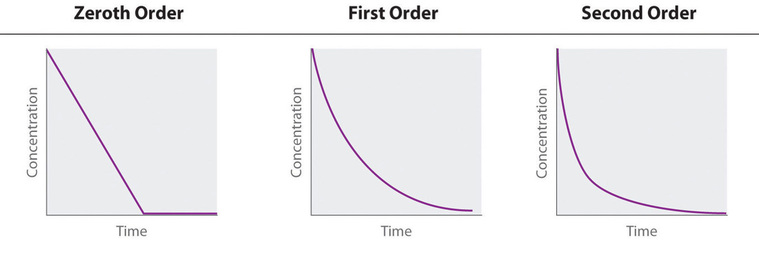
Individual reaction order = 0 : Concentration has no effect on rate. The substance must still be there but just doesn't have an effect.
Individual reaction order = 1 : Concentration is linearly related to reaction rate.
Individual reaction order = 2 : Rate is related to the square of the concentration.
Concentration vs. Rate Graphs
Individual reaction order = 0 : Concentration has no effect on rate. The substance must still be there but just doesn't have an effect.
Individual reaction order = 1 : Concentration is linearly related to reaction rate.
Individual reaction order = 2 : Rate is related to the square of the concentration.
Integrated Rate Law Graphs (Concentration vs. Time)
For zero order graphs, slope = -k or k = -slope.
For zero order graphs, slope = -k or k = -slope.
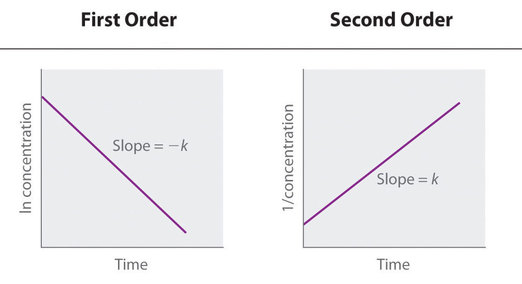
If a first order graph is converted to time vs. ln[A] instead of time vs. [A], k = -slope.
If a second order graph is converted to time vs. 1/[A] instead of time vs. [A], k = +slope.
If a second order graph is converted to time vs. 1/[A] instead of time vs. [A], k = +slope.