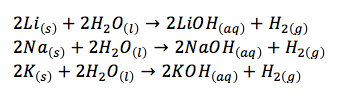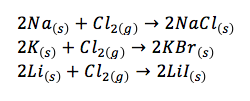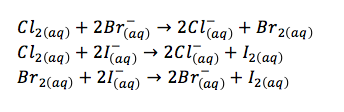3.3.1 Discuss the similarities and differences in the chemical properties of elements in the same group
Group 1: Alkali Metals
Group 1: Alkali Metals
- lithium, sodium, and potassium all have one valence electron in their outer shell
- they're very reactive and stored under liquid paraffin to prevent them from reacting with air by losing their valence electron
- their ability to readily lose an electron makes them a good reducing agent
- reactivity increases down the group because the valence electron is in successively higher energy levels and thus less energy is required to remove it
- these metals all react with water to form an alkali solution of the metal hydroxide and hydrogen gas
- lithium floats and reacts quietly, sodium melts into a ball which darts around on the surface, and the reaction with potassium generates heat which ignites the hydrogen
- alkali metals also react readily with halogens chlorine, bromine, and iodine to form ionic salts
Group 7: Halogens
- halogens react by gaining one electron to form halide ions
- because they gain an electron, they're good oxidizing agents
- reactivity decreases down the group because the outer shell is at increasingly higher energy levels and further away from the nucleus, with more shielding, so the attraction and ability to pull in an electron decreases
- chlorine is a stronger oxidizing agent than bromine, so chlorine atoms can remove the electron from bromide ions to form chlorine ions and bromine atoms
- the presence of halide ions in solution can be detected by adding silver nitrate solution
- silver ions react with halide ions to form a precipitate of the silver halide, which can be distinguished by their colour
- AgCl = white
- AgBr = cream
- AgI = yellow
3.3.2 Discuss the changes in nature, from ionic to covalent and from basic to acidic, of the oxides across period 3
Ionic to Covalent
Basic to Acidic
Basic Oxides:
Acidic Oxides:
Ionic to Covalent
- there is a change from metallic to non-metallic character across period 3
- ionic compounds are generally formed between a metal and non-metal, so the oxides formed of elements Na to Al have giant ionic structures
- covalent compounds are generally formed between non-metals, so the oxides of P, S, and Cl are molecular covalent
- the oxide of silicon, which is a metalloid, has a giant covalent structure
Basic to Acidic
- the metallic elements that form ionic metal oxides are basic
- the non-metal oxides that form covalent bonds are acidic
- aluminum and silicon show amphoteric properties, reacting with both acids and bases
Basic Oxides:
- sodium and magnesium oxides dissolve in water to form alkaline solutions with the metal bonded to a hydroxide ion
- basic oxides react with acid to form a salt and water
Acidic Oxides:
- non-metallic oxides react with water to produce acidic solutions (eg. phosphorus (V) oxide reacts with water to produce phosphoric (V) acid)