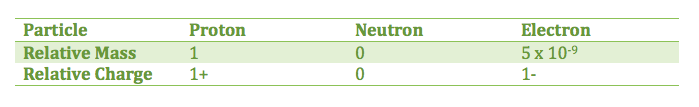
2.1.1 State the position of protons, neutrons, and electrons in the atom
- protons exist as part of the atom's nucleus
- neutrons also exist as part of the nucleus
- electrons exist around the nucleus
2.1.2 State the relative masses and relative charges of protons, neutrons, and electrons
2.1.3 Define the terms mass number (A), atomic number (Z), and isotopes of an element
- atomic number (Z): the number of protons of an atom
- mass number (M): the sum of the number of protons and neutrons
- isotope of an element: atoms of an element that have the same atomic number/# of protons but different mass numbers
2.1.4 Deduce the symbol for an isotope given its mass number and atomic number
2.1.5 Calculate the number of protons, neutrons, and electrons in atoms and ions from the mass number, atomic number, and charge
- atomic number = # protons
- # protons = # electrons (in a neutral atom)
- mass number - atomic number = # of neutrons
2.1.6 Compare the properties of the isotopes of an element
Physical Properties:
Chemical Properties:
- we compare all atoms to atoms of carbon with 6 protons and netruons
- mass of 1 atom is carbon-12
- mass it unitless or in "atomic mass units" symbol (U)
Physical Properties:
- size - no change since nucleus is such a small proportion by size of atom
- density - mass/volume. Higher density for higher isotope, lighter density for lower isotope
- diffusion rate - higher rate with lighter isotope
Chemical Properties:
- unchanged because chemical properties are reliant on the # of electrons
2.1.7 Discuss the uses of radioisotopes
3 Different Forms of Radiation:
1) Gamma (y) radiation is highly penetrating
2) Alpha (a) radiation can be stopped by a few centimeters of air
3) Beta (B) radiation can be stopped by a thin sheet of aluminum
Uses of Radioisotopes:
- many isotopes of elements are radioactive as the nuclei of these atoms break down spontaneously
- when they break down, they emit radiation which is dangerous to living things
- radioisotopes can occur naturally or be created artificially
3 Different Forms of Radiation:
1) Gamma (y) radiation is highly penetrating
2) Alpha (a) radiation can be stopped by a few centimeters of air
3) Beta (B) radiation can be stopped by a thin sheet of aluminum
Uses of Radioisotopes:
- nuclear power generation
- sterilization of surgical instruments in hospitals
- crime detection
- finding cracks and stresses in metals
- preservation of food
- carbon-14 is used for carbon dating
- cobalt-60 is used in radiotherapy
- iodine-131 and iodine-125 are used as tracers in medicine for treating and diagnosing illnesses