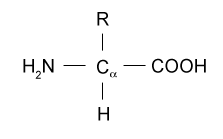
B.2.1 Draw the general formula of 2-amino acids
B.2.2 Describe the characteristic properties of 2-amino acids
- exhibits intermolecular H-bonding
- relatively high melting point
- soluble to some degree in water, dependent on R group
- R group can be polar or non-polar, + or - charge
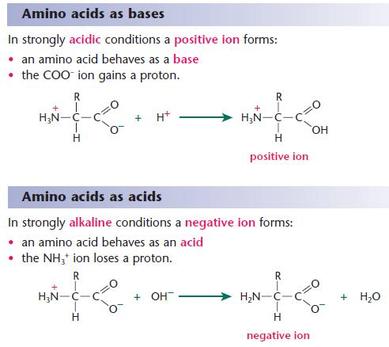
- isoelectric point (pI) is the pH at which the positive and negative charges for a given amino acid are completely balanced
- amino acids exist in solution as zwitterions
- they can also act as buffers: proton donor and acceptor are different regions of the same molecule
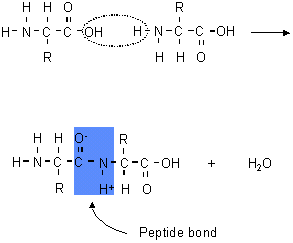
B.2.3 Describe the condensation reaction of 2-amino acids to form polypeptides
- polyamides are made of amino acid monomers joined using amide linkages (called peptide linkages in this context)
- formed through condensation reaction (dehydration synthesis)
- hydroxyl from one amino acid and a hydrogen from the other amino acid are removed and water is produced as a byproduct
- bond between a carbon and nitrogen formed as a result
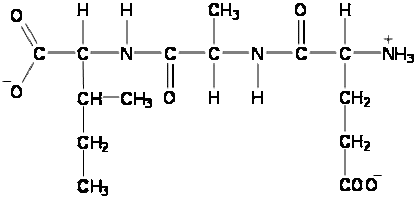
- candidates are expected to be able to draw (or split apart) up to a tripeptide of specified amino acids
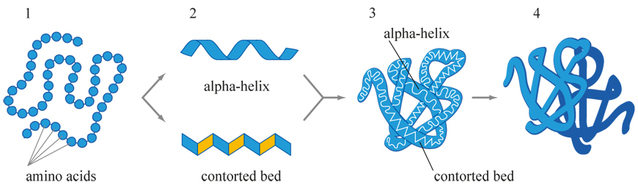
B.2.4 Describe and explain the primary, secondary (alpha-helix and beta-pleated sheets), tertiary, and quaternary structure of proteins
Protein Structure - General:
Primary Structure (1°):
Secondary Structure (2°):
Tertiary Structure (3°):
Quaternary Structure (4°):
- function follows structures
- the properties of the 3D structures are responsible for protein function
Protein Structure - General:
- generally single stable conformation (lowest energy)
- structure (folding) determined by amino acid sequence
- many noncovalent bonds stabilize
- disulfide bridges
Primary Structure (1°):
- amino acid sequence of polypeptide chain(s)
- numbered 1-X from N-terminus to c-terminus
- linear
Secondary Structure (2°):
- arrangement (folding pattern) of parts of the polypeptide backbone
- most common structures: alpha-helix and beta-pleated sheet
- other structures: turns, non-repeating (loops and coils), NOT at random
- the alpha helix: side chains project out from helix, exhibit hydrogen bonds, usually ~11 amino acids and 3 turns
- the beta sheet: adjacent chains with hydrogen bonds, rippled or "pleated" appearance, side chains alternate up and down, 5-10 amino acids per strand, 2-15 strands, cylinder or barrel shape
Tertiary Structure (3°):
- 3-dimensional arrangement of the polypeptide
- folding of the 2° structures with respect to each other
- bond structure varies among proteins
- noncovalent bonds between side chains
- cumulative effect of many weak bonds (hydrogen bonds, London forces, ionic bonds, hydrophobic interactions)
- covalent disulfide bonds are "staples" (formed AFTER folding)
Quaternary Structure (4°):
- 3-D arrangement of multiple polypeptide chains
- called "complexes"
- e.g.) myoglobin - single subunit, hemoglobin - 4 subunits
- chains fold and then assemble
- not all proteins have quaternary structure
B.2.5 Explain how proteins can be analyzed by chromatography and electrophoresis
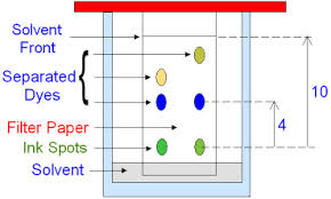
Paper Chromatography
Chromatography Procedure:
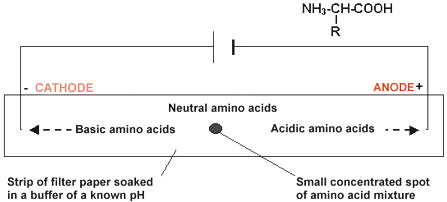
Amino Acid Electrophoresis
E.g.) Gly has pI=6.0
Electrophoresis Procedure:
Paper Chromatography
- good for separating hydrophilic substances
- different components (amino acids) will be separated due to differential solubility in the solvent
- stationary phase --> paper
- mobile phase --> solvent
- the mobile phase travels through the stationary phase by capillary action
- the sample will only move up the chromatogram while dissolved in the mobile phase
- the more soluble a sample or component of the sample, the more time it spends in the mobile phase and the further it will travel
- often results are reported as Rf values (retention factor), which is the ratio of (distance sample travels)/(distance solvent front travelled)
Chromatography Procedure:
- must remove paper from tank BEFORE solvent front goes off the end of the paper
- remove paper and mark the solvent front
- dry the paper
- amino acids are transparent so they need a treatment to visualize them
- standard visualization is to spray with ninhydrin, which makes amino acids purple/blue (except for proline which will turn yellow/orange)
- measure travel distance to centre of spots
- calculate Rf, compare to knowns
Amino Acid Electrophoresis
- separation will be based on electric charge of amino acids
- to work out amino acid composition of proteins, primary structure must be broken through hydrolysis, which requires 6 mol/L HCl, 110°C, and 1-3 days to break the peptide bonds
- as previously stated in B.2.2, amino acids each have an isoelectric point (pI)
- if the amino acid has -COO-, pI will be at lower pH
- if the amino acids has -NH2, pI will be at higher pH
- substances move towards oppositely charged ends of an electric field when placed in gel or paper saturated with a buffer of a certain pH
- the further the buffer pH is from the pI, the further towards an electrode the amino acid will move
E.g.) Gly has pI=6.0
- if paper or gel saturated with buffer of pH 6.0, pI=pH so gly has no overall charge so it won't move
- if buffer is pH 8.0, pH>pI so gly will be negatively charged and move towards the positive end of the chamber
- if buffer is pH 4.0, pH<pI so gly will be positively charged and move towards the negative end of the chamber
Electrophoresis Procedure:
- apply mixture of amino acid to paper/gel with each end in specific pH buffer, and electrodes at each end
- apply voltage and develop with ninhydrin (or similar) to visualize amino acid
- measure distances migrated (and direction) and compare to known values for particular pH
B.2.6 List the major functions of proteins in the body
Fibrous Proteins
Examples:
2. Silk Fibroin
Globular Proteins
- proteins are macromolecules that make up ~50% of non-water body mass
Fibrous Proteins
- elongated
- one type of 2° structure is predominant (simple)
- usually are assemblies of many of the same protein
- very structural (protective, connective, motive)
- e.g.) keratin, collagen, elastin, silk
Examples:
- Alpha keratin
- coiled coil: 2 alpha helices with hydrophobic stripe
- microfibrils --> macrofibrils --> cells in hair
2. Silk Fibroin
- fibroin + gum protein = silk
- series of beta sheets
- gly-ser-gly-ala repeat of amino acids
- R groups interlock
- very strong
Globular Proteins
- compact, spheroidal
- more than 1 type of 2° structure, variety of motifs and domains
- single or multichain, solitary or assemblies (same or different proteins)
- highly diverse (enzymes, transport proteins, receptors, immunoproteins)
- e.g.) lysozyme, insulin receptor, hemoglobin, antibodies