6.2.1 Describe the kinetic theory in terms of the movement of particles whose average energy is proportional to temperature in Kelvins
- the average kinetic energy of a sample of gas particles is directly proportional to the temperature in Kelvin of the sample
6.2.2 Define the term activation energy, Ea
- the minimum increase in potential energy of a system required for molecules to react
- minimum energy required for a chemical reaction to occur
6.2.3 Describe the collision theory
For a chemical reaction to take place, particles must collide with:
1) The proper collision geometry or collision orientation
2) Sufficient energy for the reaction to take place
Reaction rate depends on:
1) Frequency of collisions between reactants
2) Proportion of effective collisions
The rate is not linear because: The initial rate is highest because reactant concentration is highest at the beginning and therefore effective collision frequency is highest then. As the reaction proceeds, reactants are converted to products and reactant concentration drops. Thus, collision frequency decreases and rate decreases.
For a chemical reaction to take place, particles must collide with:
1) The proper collision geometry or collision orientation
2) Sufficient energy for the reaction to take place
Reaction rate depends on:
1) Frequency of collisions between reactants
2) Proportion of effective collisions
The rate is not linear because: The initial rate is highest because reactant concentration is highest at the beginning and therefore effective collision frequency is highest then. As the reaction proceeds, reactants are converted to products and reactant concentration drops. Thus, collision frequency decreases and rate decreases.
6.2.4 Predict and explain, using the collision theory, the qualitative effects of particle size, temperature, concentration, and pressure on the rate of reaction
Particle Size:
Concentration/Pressure:
Temperature:
Particle Size:
- decreasing particle size means increasing the surface area of a given sample
- increases reaction rate
- you can only increase surface area of a solid
- only particles on the surface of a solid are available for collisions
- smaller particles have a greater surface over which the reaction can take place
- thus increased surface area means increased collision frequency
- a higher frequency of collisions means higher frequency of effective collisions
- therefore the rate of reaction will be higher
Concentration/Pressure:
- increased concentration increases rate
- can only change the concentration of an aqueous solution or gaseous reactant
- increasing concentration of a reactant increases # of particles within a given volume
- therefore frequency of reactant-reactant collisions will be higher
- therefore frequency of effective collisions will be higher
- thus, rate of reaction will be higher
- the same explanation applies to increasing pressure of a gas by decreasing volume of the container
Temperature:
- the average kinetic energy of a sample of gas particles is directly proportional to the temperature in Kelvin of the sample
- increasing temperature primarily increases reaction rate because an increase in temperature increases the average kinetic energy of the reactant particles
- therefore, when reactant particles collide with each other, there is a higher probability that they will have sufficient energy for an effective collision (kinetic energy is sufficient to surpass the activation energy)
- this will generate a higher proportion of effective collisions, and the reaction rate will be higher
- temperature also affects collision frequency since particles with higher energy will collide more frequently, also increasing effective collision frequency and thus increasing reaction rate
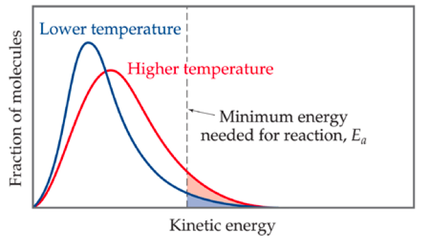
6.2.5 Sketch and explain qualitatively the Maxwell-Boltzman energy distribution curve for a fixed amount of gas at different temperatures and its consequences for changes in reaction rate
- a Maxwell-Boltzmann distribution shows the energy distribution of gas particles in a given sample
- the x axis represents the amount of energy and the y axis and area under the curve represent the # of particles
- the peak of the graph shows most particles having moderate energies, with some particles having very high energies and a few particles having very low energies
- at a lower temperature, the particles to the right of the activation energy (Ea) boundary have sufficient energy to react
- at a higher temperature, due to an increase in average kinetic energy of the particles, there are more particles that have sufficient energy to react
- the reason why the area under the curve stays constant is because we are not losing particles and decreasing the number of them, but rather merely changing how much energy these particles have - this shifts the energy distribution, not the # of particles
6.2.6 Describe the effect of a catalyst on a chemical reaction
- a catalyst is a substance that increases the rate of a reaction without being consumed
- one kind of catalyst provides a surface for reactions to happen on
- in this case, collision geometry will be affected increasing chances of an effective collision
- also the absorption may place a strain on bonds that need to be broken
- this will decrease the activation energy for the reaction
- the amount of energy required for a reaction to take place is lower for a reaction when a catalyst is present because a catalyst creates an alternative reaction pathway
- a lower Ea means more particles have sufficient energy to react, thus proportion of effective collisions will be higher
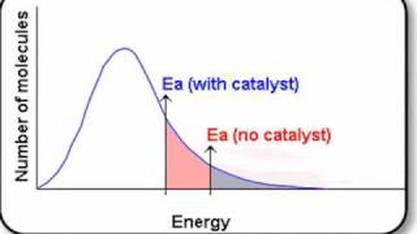
6.2.7 Sketch and explain Maxwell-Boltzmann curves for reactions with and without catalysts
- because a catalyst lowers the activation energy required for a reaction to take place, the energy distribution of the particles won't need to change like with a change in temperature
- rather, the minimum energy boundary that the particles need to "cross", as shown through the lines with arrow in the graph below, is changed so that more of the curve is past the boundary
- thus, it means that more of the particles have sufficient energy