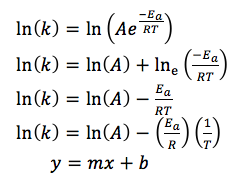16.3.1 Describe qualitatively the relationship between the rate constant (k) and temperature (T)
Increasing the temperature of a reaction increases the rate of reaction, and the relationship between k and T is shown in the form of the Arrhenius equation.
Increasing the temperature of a reaction increases the rate of reaction, and the relationship between k and T is shown in the form of the Arrhenius equation.
16.3.2 Determine activation energy (Ea) values from the Arrhenius equation by a graphical method