7.1.1 Outline the characteristics of chemical and physical systems in a state of equilibrium
A system at equilibrium:
1) The system must be closed
2) The system must have a constant temperature
3) No macroscopic changes are occurring (eg. changes in colour, volume, pH, etc.)
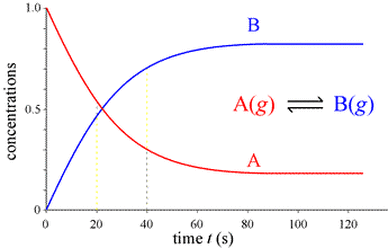
4) The rates of the forward reaction and reverse reactions are equal (A + B <--> C + D)
5) Equilibrium in a given system can be approached from both directions
A system at equilibrium:
1) The system must be closed
2) The system must have a constant temperature
3) No macroscopic changes are occurring (eg. changes in colour, volume, pH, etc.)
4) The rates of the forward reaction and reverse reactions are equal (A + B <--> C + D)
5) Equilibrium in a given system can be approached from both directions
|
Initially, the reactants will be consumed rapidly and the formation of products will occur slowly (possibly even zero). As the reaction proceeds, the net concentration of reactant that needs to be converted drops, so the reaction rate drops, and the formation of products occurs faster. The closer to equilibrium the system becomes, the smaller the difference in rate for the forward and reverse reactions, so the net change in concentration for all reactants becomes smaller.
|